

The success of dental implants hinges on a multitude of factors, including both local and systemic conditions such as bone quality, systemic diseases, implant design, and notably, implant surface characteristics. Surface treatments play a pivotal role in enhancing osseointegration—the direct structural and functional connection between living bone and the implant surface. Traditional methods like acid etching, sandblasting, and plasma spraying have been extensively studied for their ability to modify surface roughness and topography, thereby improving cell adhesion and proliferation. Recent advancements have introduced bioactive coatings, such as collagen, bone morphogenetic proteins (BMPs), and hyaluronic acid, which further promote osteogenic differentiation and accelerate bone healing. Emerging technologies, including femtosecond and picosecond laser treatments, offer precise control over surface microstructures, enhancing antibacterial properties and osteointegration potential. Additive manufacturing techniques, like selective laser melting, allow for the creation of porous structures that mimic natural bone architecture, facilitating vascularization and cellular infiltration . However, challenges remain regarding the long-term stability and durability of these surface modifications, especially in patients with compromised bone quality or systemic conditions. Therefore, ongoing research is essential to optimize surface treatment strategies, ensuring enhanced implant performance and improved patient outcomes in dental implantology.
Key words: machining, acid etching, laser ablation, electropolishing, plasma spraying, anodization, electrophoretic deposition, biomimetic coating
Dental implants have become a leading solution
for partial and complete tooth loss, offering
superior aesthetics, function, and long-term
outcomes compared to traditional prosthetics.
Titanium is the preferred implant material due
to its strength, biocompatibility, and resistance
to degradation. Factors such as implant design,
surface topography, and bone quality influence this process.1 To enhance osseointegration,
various surface modification techniques
like
sandblasting, acid etching, plasma
spraying, and anodization are used to improve
surface roughness and cellular response.
Bioactive coatings (e.g., calcium phosphate,
hydroxyapatite) and nanotechnology further
boost bone regeneration by mimicking natural
bone structure.
Incorporating
biological
molecules like BMP-2 and collagen shows
promise in enhancing integration, especially
in complex cases. Despite high success rates,
issues like bacterial colonization and peri
implant bone loss persist, emphasizing the need
for ongoing research into safer, more effective,
and personalized implant solutions.2
Implant surface treatments play a pivotal role
in enhancing osseointegration and clinical
success. To enhance osseointegration, various
surface modifications are required. These
surface modifications are generally classified
into three major categories: subtractive methods,
additive methods, and biochemical methods.[3] Each approach offers distinct mechanisms
for improving implant-tissue interaction,
and numerous studies have explored their effectiveness in promoting early healing, bone
formation, and long-term stability.

Machining (Turning)
Machining was the first surface treatment used
for dental implants, introduced in the 1970s by
Brånemark. This process involves mechanical
shaping through turning or milling, producing
a
smooth, polished surface with limited
topographical features. Though appearing flat
to the naked eye, SEM analysis reveals fine
grooves with surface roughness values typically
ranging from Sa 0.5–1.0 μm and Ra 0.8–1.2 μm.
Due to this low roughness, machined surfaces
offer limited bone interlocking and slower
osseointegration.1
Acid etching
Acid etching is a widely used subtractive
technique that enhances implant surface
roughness by exposing titanium to strong acids
such as HCl, H2SO4
, HNO3
, or HF. This process
removes the oxide layer, revealing fresh, reactive
titanium and creating micro-pits (0.5–2 μm) that
promote protein adsorption, cell attachment,
and osteoblast activity.4 The increased surface area and microroughness (Ra ~1.5 μm) improve
bone-to-implant contact and osseointegration.
Dual acid etching (DAE)
It is a surface modification technique that involves
treating titanium implants with a combination of
strong acids. This process creates a micro-rough
surface with submicron topography, enhancing
surface area, protein adsorption, and osteoblast
attachment.5 The resulting surface roughness
(Ra ~1–2 μm) improves bone-to-implant contact
and mechanical interlocking, accelerating
osseointegration. DAE also alters surface
chemistry, increasing biocompatibility and
facilitating early bone healing. In some cases,
fluoride ions retained on the surface provide
added antibacterial and osteoconductive
benefits. Compared to smoother or single-etched
surfaces, DAE-treated implants demonstrate
higher stability, faster integration, and improved
clinical outcomes.6
Laser ablation (Laser peening)
Laser surface modification enhances dental
implants by creating precise micro/nano
textures using lasers like Nd:YAG, Er:YAG,
CO2
, or femtosecond types. Techniques such as
laser ablation improve roughness, wettability,
biocompatibility, and bacterial resistance.7
Nanosecond lasers reduce heat damage,
ensuring accuracy. On titanium, lasers form
oxide layers; on zirconia, they enhance bonding.
Products like Laser-Lok® use microchannels
for better soft tissue integration. However, the
method requires expertise and strict safety
measures.8
SLA (Sandblasted, Large-grit, Acid
etched)
The SLA surface is a widely used dental implant modification designed to enhance
osseointegration, introduced by the Straumann
Institute in 1997. The SLA process involves
large-grit sandblasting—typically with Al2O3
,
TiO2
, or HA—to create a macro-rough surface
(20–40 μm craters), followed by acid etching with
HCl and H2SO4 to form micro-pits (0.5–3 μm)
and nanoscale features. This dual treatment
increases surface area, surface energy, and
promotes bone cell adhesion and proliferation.
The resulting surface has a roughness (Ra)
around 1.5 μm and a thickened oxide layer,
enhancing protein adsorption, bone-implant
contact (BIC), and osseoconductivity. Its isotropic
texture and improved hydrophilicity support early
implant loading and better clinical outcomes.9
In 2005, the SLActive surface—a chemically
modified SLA with enhanced hydrophilicity—
was introduced, offering faster and more reliable
osseointegration.
Electropolishing
Electropolishing, also known as anodic
polishing, is an electrochemical process used
to smooth and passivate metal surfaces by
selectively removing surface irregularities. Using
a concentrated acid electrolyte (typically sulfuric
and phosphoric acid), the process reduces
roughness, enhances corrosion resistance,
and improves biocompatibility. It also removes
microburrs and contaminants, forming a clean,
passive oxide layer ideal for medical and
pharmaceutical applications.10
Plasma spraying
It is a thermal coating technique in which
powdered materials are melted using a high
temperature plasma jet and sprayed onto
implant surfaces. This method allows precise control over coating thickness, porosity, and
composition, enhancing implant performance
and osseointegration. Titanium plasma
spraying (TPS) creates a rough surface with
micro-irregularities that increase surface area
and improve mechanical interlocking with
bone, aiding initial stability, especially in soft
bone. Hydroxyapatite (HA) coatings, applied
similarly, form a bioactive layer that promotes
direct bone bonding and supports osteogenic
cell attachment.11 While both coatings enhance
osseointegration, concerns over titanium wear
particles from earlier TPS surfaces have led to
a shift toward safer, moderately rough surfaces.
Anodization
Anodization is an electrochemical process that
enhances the surface of titanium dental implants
by forming a controlled oxide layer. The titanium
acts as the anode in an acidic electrolyte solution,
typically containing sulfuric, phosphoric, and
hydrofluoric acids, under voltages of 1–300
V and temperatures of 18–38°C. This process
creates an oxide layer with varying thickness
(1–10 μm) and roughness (Ra>2 μm), improving
corrosion resistance, wear resistance, and early
osseointegration. Anodized surfaces increase
protein adsorption and cell attachment.12
Electrophoretic deposition (EPD)
EPD is an electrochemical technique used
to apply uniform coatings on conductive
implant surfaces by moving charged particles
in a suspension under an electric field. It is
commonly used to deposit bioactive materials
like hydroxyapatite (HA), which enhances cell
adhesion and osseointegration.13
Biomimetic coating
These coatings aim to replicate the natural extracellular matrix (ECM) of bone, promoting
cellular activities essential for successful
osseointegration. It has been shown that
such biomimetic coatings are more soluble
in physiological fluids and resorbable by
osteoclastic cells such as dentin materials.[10]
Agents used:

Immobilization of growth factors
One effective approach to enhance
osseointegration and promote bone regeneration
around dental implants involves the surface
immobilization of growth factors. These biological
molecules, such as bone morphogenetic proteins
(BMPs) and platelet-derived growth factors
(PDGFs), play crucial roles in regulating cell
proliferation, differentiation, and extracellular
matrix synthesis.14 Various techniques such
as physical adsorption, covalent attachment,
and affinity-based methods—can be used to
anchor growth factors onto implant surfaces.15
Once immobilized, these growth factors interact
with specific cell surface receptors, triggering
intracellular signalling pathways that support
cellular activity and accelerate bone tissue
formation.
Peptide Functionalization
To enhance the biological performance of
implants, their surfaces can be modified
with biomolecules. This involves the physical
or chemical attachment of substances like
proteins, peptides, or components of the
extracellular matrix. Techniques such as self
assembled monolayers, plasma polymerization,
and layer-by-layer deposition allow precise
control over biomolecule immobilization. These
functionalization methods support specific
cellular interactions, influencing cell adhesion,
growth, differentiation, and matrix production,
thereby accelerating healing and improving
osseointegration.16
Protein Coating
Protein coatings on dental implants aim to
replicate the natural extracellular matrix and
provide a bioactive surface that enhances cell
attachment and proliferation. Type I collagen, the most abundant protein in bone, is frequently
used due to its osteoconductive properties and
its ability to promote osteoblast attachment.17
When coated on the implant surface, it acts as
a scaffold for new bone formation, supporting
both osteogenic cell recruitment and matrix
mineralization. Fibronectin and laminin,
other key matrix proteins, play crucial roles in
epithelial cell adhesion and connective tissue
formation. These coatings also help establish
a strong mucosal seal around transmucosal
implants, which is vital for preventing bacterial
ingress and peri-implantitis.18
Enzyme Immobilization
It is a biochemical surface treatment that involves
binding biologically active enzymes to the
implant surface to influence mineralization and
tissue remodeling. Alkaline phosphatase (ALP) is
an osteogenic enzyme that plays a key role in the
mineralization of bone matrix. By immobilizing
ALP on the implant surface, calcium phosphate deposition is enhanced, promoting rapid
formation of a mineralized layer at the implant
bone interface.19 This contributes to stronger
and faster osseointegration. Another enzyme,
lysyl oxidase (LOX), catalyzes cross-linking
in collagen fibers and strengthens connective
tissue attachments, which is particularly
beneficial in soft tissue integration. These
enzymes are typically stabilized on the surface
through chemical crosslinking or entrapment
methods, ensuring sustained biological activity
post-implantation.4
Antimicrobial Agent Incorporation
To reduce bacterial colonization and prevent
peri-implantitis, antimicrobial agents are
incorporated on implant surfaces. Common
approaches include controlled-release antibiotic
coatings (e.g., vancomycin, gentamicin),
antimicrobial peptides (AMPs) that also aid
healing, and silver nanoparticles (AgNPs) known
for broad-spectrum, long-lasting effects. These
agents can be applied through adsorption,
chemical bonding, or embedded in coatings,
providing localized infection control—especially
beneficial for high-risk patients.20
Recent Advances in Implant Surface
Treatment
Continuous advancements in dental
implant surface engineering have led to the
development of novel approaches aimed at
improving the biological response, enhancing
implant stability, reducing healing time, and
minimizing complications.21 These techniques
are increasingly moving beyond traditional
methods to incorporate biologically active,
smart, and patient-specific strategies. The
following sections outline the most significant
recent developments.
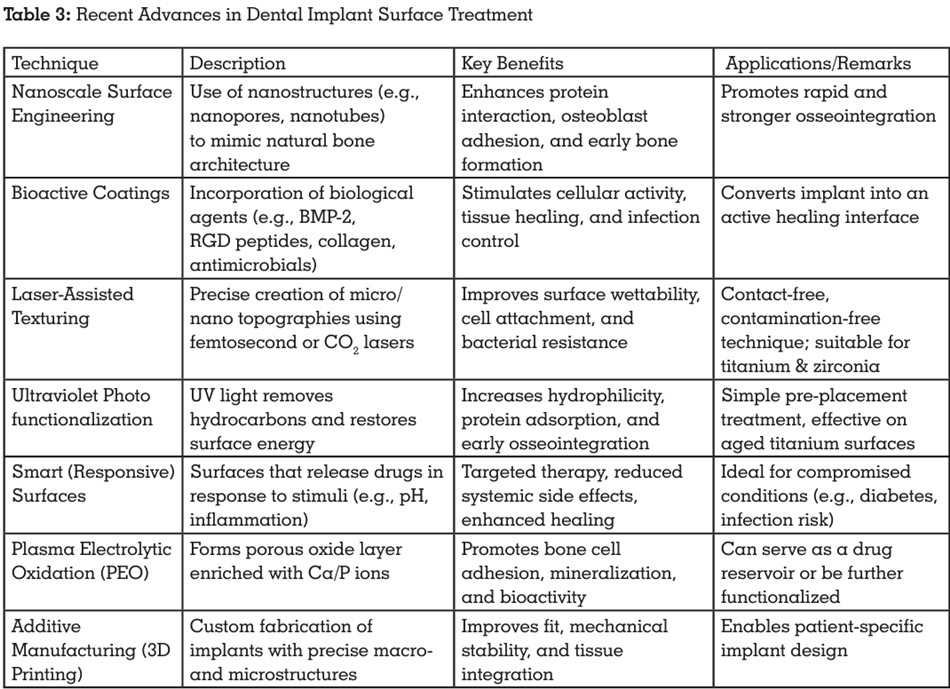
Nanotechnology has enabled the creation of implant surfaces with structures that closely resemble the nanoscale features of natural bone.22 By introducing nanostructures—such as nanopores, nanotubes, and nanorough surfaces—cellular activity is significantly enhanced. These surfaces support improved protein interaction, encourage osteoblast adhesion and proliferation, and promote early bone regeneration, thereby improving the quality and speed of osseointegration.23
Functional Bioactive Coatings
Recent strategies focus on applying biologically
active molecules to the implant surface to elicit
targeted cellular responses. These coatings
transform the implant from a passive device into
a biofunctional interface that actively supports
healing and integration. These include:
Laser-Assisted Surface Texturing
Laser surface modification is a precise and
efficient method that uses focused laser energy
to create detailed micro- and nano-topographies
on implant surfaces. Different types of lasers (e.g., femtosecond, CO2
, nanosecond lasers) are used
to create clean, uniform textures without physical
contact or contamination.27 These modified
surfaces show improved wettability, better cell
attachment, and enhanced integration with both
soft and hard tissues. Additionally, the textured
patterns may help resist bacterial colonization
and reduce the risk of peri-implantitis.28
Ultraviolet Photofunctionalization
It is a surface treatment technique used to
enhance the hydrophilicity and biological
activity of titanium dental implants. Over time,
implant surfaces can accumulate hydrocarbon
contaminants, making them hydrophobic and
less receptive to cellular attachment. Exposure to ultraviolet (UV) light, particularly UV-C,
removes these organic residues and activates
the titanium oxide layer, restoring surface energy
and converting the implant to a superhydrophilic
state.29 This improves protein adsorption,
promotes osteoblast adhesion, and accelerates
early osseointegration. The treatment is simple,
safe, and non-invasive, typically performed
shortly before implant placement to optimize
tissue response and clinical outcomes.30
Responsive (Smart) Implant Surfaces
Smart implant surfaces are designed to respond
to physiological conditions in real time. These
surfaces can release therapeutic agents—such
as antibiotics or anti-inflammatory compounds— when triggered by specific stimuli like changes
in pH or the presence of inflammation.31 This
targeted delivery approach supports local
treatment at the implant site while minimizing
systemic side effects, and enhances healing
especially in compromised conditions.32
Plasma Electrolytic Oxidation (PEO)
It is a technique that generates a porous and
chemically active oxide layer on titanium
implant surfaces. These oxide layers are often
enriched with calcium and phosphorus to
enhance bioactivity.33 PEO-treated surfaces
improve osseointegration by supporting bone
cell attachment and mineral deposition.
Additionally, their porous architecture can serve
as a reservoir for drug delivery or further surface
functionalization.34
The integration of 3D printing in dental
implantology has made it possible to design
implants with complex, customized geometries
tailored to individual patient anatomy. This
technology allows precise control over macro
and microstructural features during fabrication,
including built-in porosity and surface texturing.
As a result, these implants achieve better fit,
mechanical stability, and tissue integration,
making them especially useful for patients with
challenging anatomical conditions.35
The choice of implant surface treatment plays
a critical role in clinical success and must
be tailored to each patient’s biological and anatomical condition.12 Factors such as bone
quality, systemic health, esthetic zone placement,
and risk of infection significantly influence the
selection of surface modifications. A practical
workflow helps guide clinicians in choosing the
most appropriate implant surface for predictable
outcomes.
In the following table, a simplified clinical
decision-making guide is presented to help
clinicians align patient profiles with surface
treatments, considering both clinical and
biological
demands.36,37,38,39,40,41,42
clinical implications.

Surface modifications of dental implants are
a cornerstone of modern implantology, aiming
to enhance osseointegration by modulating
the implant–bone interface at microscopic and
nanoscopic levels. The primary goal of these
treatments is to stimulate early cellular responses
and promote robust bone formation around the
implant, particularly during the critical healing
period.
Conventional techniques such as acid etching
and sandblasting create a micro-rough surface,
which increases the surface area and facilitates
mechanical interlocking. These methods also
improve protein adsorption and osteoblast
attachment, leading to a stronger bone-to
implant contact (BIC) and enhanced stability
during early healing phases43,44. Sandblasted,
large-grit, acid-etched (SLA) surfaces and
their hydrophilic variants like SLActive have
been widely adopted for their ability to reduce
healing time and support immediate loading
protocols9,36.
Plasma-sprayed coatings, notably those using
titanium plasma spray (TPS) and hydroxyapatite
(HA), are additive methods that increase implant surface area and bioactivity. These coatings aim
to mimic the mineral component of bone and
serve as a scaffold for osteoblasts. However,
challenges such as delamination, variable
thickness, and long-term mechanical stability
persist, especially under functional load over
time11,44.
Recent innovations have turned attention
toward nanostructured and bioactive surfaces.
These surfaces can be engineered to include
growth factors, peptide sequences (e.g.,
RGD), type I collagen, and even extracellular
matrix proteins like fibronectin or laminin15,17,25.
Such functionalization mimics the natural
bone environment, accelerating osteogenic
differentiation, improving tissue integration,
and significantly enhancing the potential for
early implant loading. These strategies not only
promote osseointegration but also support soft
tissue healing and mucosal sealing, thereby
reducing the risk of peri-implantitis16,18.
Further, laser-assisted surface modification
techniques using femtosecond or nanosecond
lasers allow precise control over implant
topography without introducing contaminants.
These methods improve wettability, promote
early cell attachment, and exhibit promising
antibacterial properties. Products like Laser
Lok® utilize microchannels for better soft tissue
integration, contributing to long-term implant
success27,28.
Zirconia implants have emerged as a valuable
alternative to titanium due to their superior
esthetics and biocompatibility, especially in the
anterior maxilla. However, their relatively lower
fracture toughness and limited osseointegration
capacity—owing to their bioinert surface—
necessitate further optimization of surface
treatments such as laser texturing, UV
photofunctionalization,
and bioactive coatings46,48. Although short-term studies show
promising outcomes, the long-term clinical data
on zirconia implants remain limited.
Another area of concern is the lack of standardized
protocols for surface modifications. With the
variety of methods—ranging from subtractive
approaches like sandblasting to biochemical
coatings with proteins or antimicrobial agents—
there is often variability in clinical outcomes,
particularly when treating compromised patients
(e.g., those with diabetes, osteoporosis, or poor
bone quality)47,49. This inconsistency underscores
the importance of personalized, case-specific
treatment strategies that consider the patient’s
systemic health, bone density, and esthetic
requirements.
Moreover, smart surfaces, capable of drug
release in response to inflammation or pH
changes, and PEO-treated surfaces enriched
with calcium and phosphate ions, are paving the
way for implants that can respond dynamically
to biological conditions. When integrated with
3D printing, clinicians can create implants
tailored to individual anatomical and biological
profiles, further enhancing clinical outcomes in
complex cases31,32,35.
While traditional methods continue to play an
important role, the integration of biomimetic
design, nanotechnology, and biochemical
functionalization is reshaping the future of
implantology. The challenge remains to validate
these techniques through long-term clinical trials
and to develop consensus-driven guidelines for
consistent, safe, and effective application across
diverse patient populations.
Implant surface treatment is a critical
determinant of successful osseointegration and
long-term implant stability. Techniques such as sandblasting, acid etching, anodization, plasma
spraying, and bioactive coatings have proven
effective in enhancing surface roughness,
wettability, and biological response.48 Advanced
modifications, including nano-structuring and
incorporation of bioactive agents like BMP
2, hydroxyapatite, and collagen, have shown
significant improvements in bone-to-implant
contact and early integration.49 Emerging
interest in zirconia implants also highlights the
role of material-specific surface treatments in
expanding clinical applications.50
However,
standardization of protocols and long-term
clinical validation remain necessary.
Overall, innovations in surface modification
continue to improve implant outcomes, offering
better integration and durability—especially
crucial in complex or compromised clinical
conditions.