Dr Prasanth V
Editor
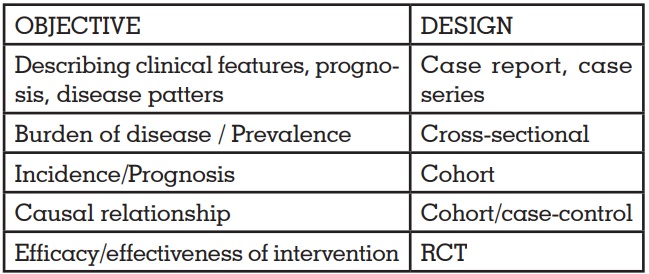
RESEARCH DESIGNS – A Quick Guide
EXPERIMENTAL DESIGN CONCEPT
Two or more groups are involved in the study. One group
is the intervention group (new treatment/drug) and the
other forms the Control group (standard/
placebo). Investigator
decides in the choice
of intervention.
This includes randomized and nonrandomized control trials.

OBSERVATIONAL DESIGN CONCEPT
Investigator stands apart
from the events taking
place from the study,
simply observe and record.

DESCRIPTIVE DESIGN CONCEPT
First scientific toe in water in new areas of inquiry. Good
descriptive study answers five ‘W’ questions, who, why,
what, when and where and the 6th one. ‘so what’. They
describe or estimate a phenomenon in terms of ‘person’,
‘place’ and ‘time’. This design generate ‘hypothesis’ (no
relationship can be proved) which can be tested using
‘analytical’ type of designs.

ANALYTIC DESIGN CONCEPT
Used to test hypothesis
concerning relationship
between a suspected
risk factor and an outcome and to measure
the magnitude of the
association and its
statistical significance.
They are called natural
experiments.

CASE REPORT
Observational and descriptive. It’s a careful detailed report
of the profile of a single patient or case. Eg- Case of 40
year old woman developing pulmonary embolism within
5 weeks of oral contraceptive usage in the year 1961.
CASE SERIES
Observational and descriptive. Series of homogenous
cases are studied and described. Eg – A review of all
patients diagnosed as endometrial carcinoma and look
for history of hormone replacement therapy.

CROSS SECTIONAL STUDY (Snapshot studies, Prevalence Studies) Observational and descriptive (can be
analytical also if there is comparison of groups based on
exposure or outcome). These studies provide information concerning a situation (exposure and outcome/disease) at
a given time and hence provides information concerning
a situation at a given point time only. Not feasible to study
rare conditions and not ideal for studying rare exposures.
Eg – Prevalence of perceived stress among students of
various Medical Colleges under KUHS.

ECOLOGIC STUDY
Observational and descriptive
(can be analytical also). Here the
unit of study is not individuals,
but populations. Exposure and
outcome obtained from different
groups or populations are compared. Since ecologic studies
refer to ‘populations’ and NOT
‘individuals’, it is not possible link
the exposure and outcome in an
individual level (ecologicfallacy).
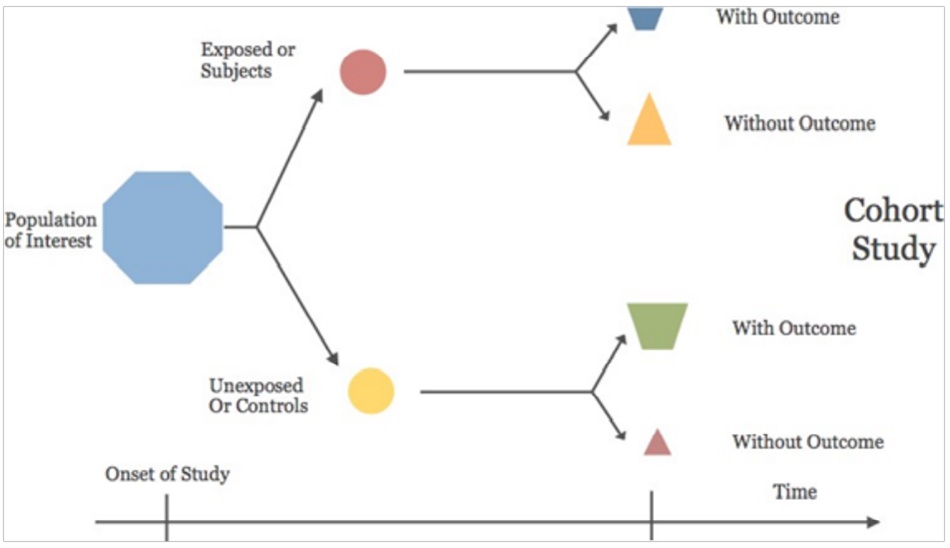
COHOHT STUDY (Longitudinal / Incidence Studies)
Group of persons (Cohort) are identified based on their
exposure. They should be free of disease/outcome of interest, but differ in exposure selected. They are followed
identically until they develop outcome/disease. Incidence
of disease and risk calculated.
Eg–Risk of passive smoking among pregnant women on
low birth weight of babies.
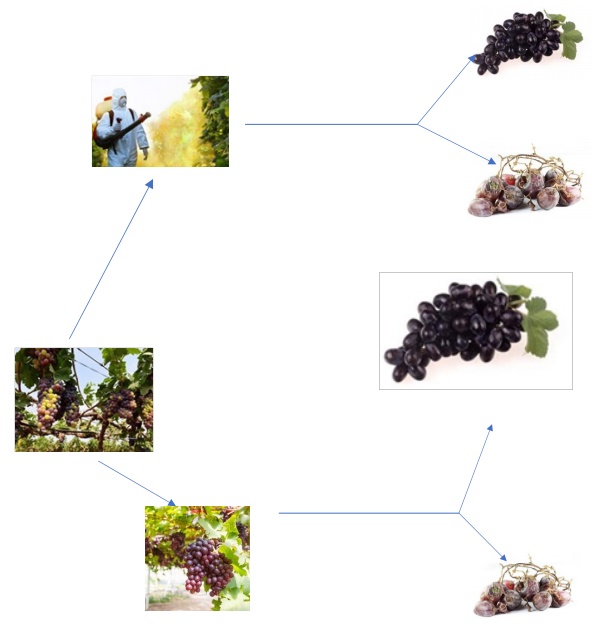
Easy Learning - In one vineyard pesticides are routinely
applied and in other vineyard it’s not applied normally.
There was a hypothesis that, the spoiled grapes were less
in the first vineyard. To verify that, compare the amount
of good and spoiled grapes at the time of cultivation
obtained from both farms.
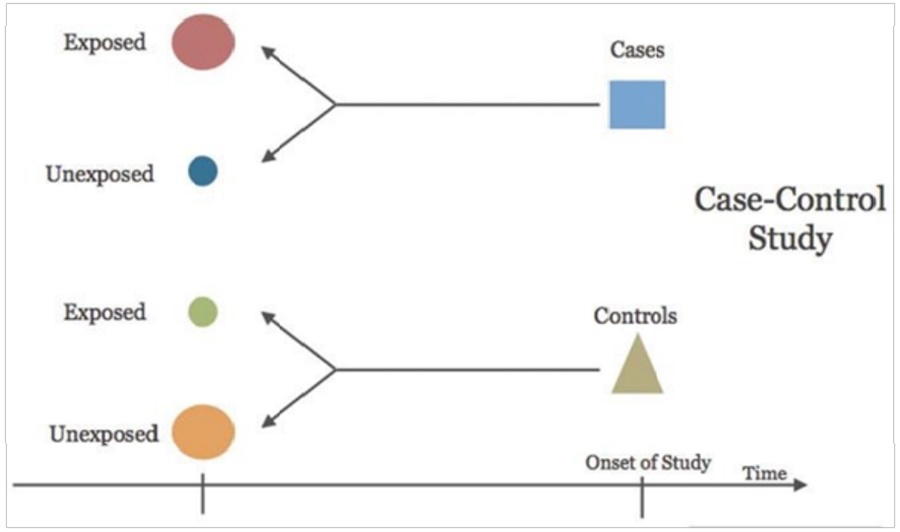
CASE-CONTROL STUDY
It’s a retrospective study design because group of subjects with disease/outcome of interest (cases) and group
of subjects without disease/outcome of interest (control)
are identified first. Cases and controls are investigated
for presence or absence of hypothesized risk/exposure
factors. Analysis in this type of studies are done by calculating Odds Ratio (OR).
Eg – Is post-menopausal hormone replacement therapy
associated with increased risk of endometrial cancer?
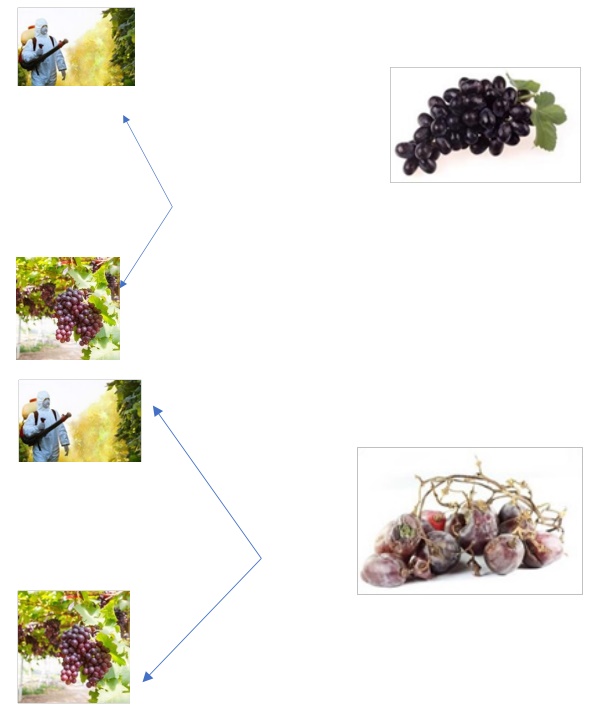
Easy Learning - Just the reverse of what we saw in cohort
study. Here we start with identification of spoiled grapes
(case) and good grapes (control). History of pesticide
exposure in both groups are investigated and revealed.
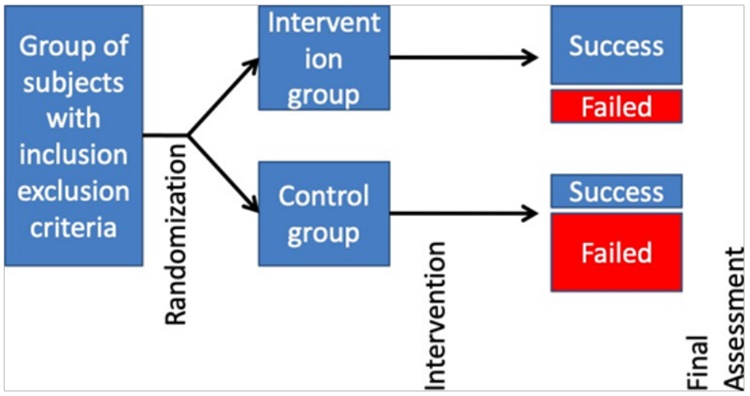
RANDOMIZED CONTROL TRIALS (RCT)
It offers highest form of evidence regarding association
and causation. The word ‘control’ means that observations are conducted under controlled circumstances in
which the researcher manipulates the conditions. This
‘controlled’ experiment can be randomized or non-randomized. ‘Randomization’ ensures that the assignment
of study participants into ‘study’ and ‘control’ groups are
done by ‘chance’ alone. This is almost similar to Cohort
study, but the intervention is decided by the investigator.
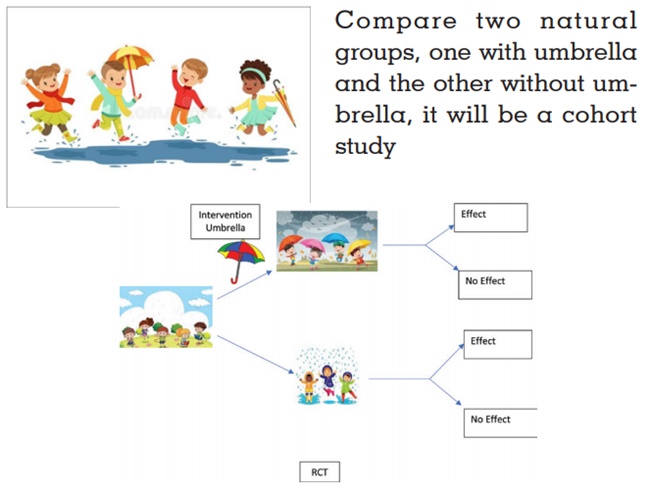
Easy Learning - We want to study the effect of rain on the
health of children If observe children standing in the rain
with and without umbrellas whom we divide into two groups
(one with umbrella and the other without umbrella) and
study the effects, then that will be a cohort study, a natural
experiment. If the investigator divide children standing
in rain randomly into two groups, give umbrellas to one
group (study group) and no umbrellas to the other group
(control group) and both groups are then compared, it
will be a RCT.
Parallel Design, Cross over design, Factorial Design,
Multi-centric trials, Mega trials and Split half designs
(split mouth, split eye etc) are various types of clinical
trials. According to purpose of research, trials can be
therapeutic trials, prophylactic trials, safety trials and
risk factor trials.
HIERARCHY OF RESEARCH DESIGNS
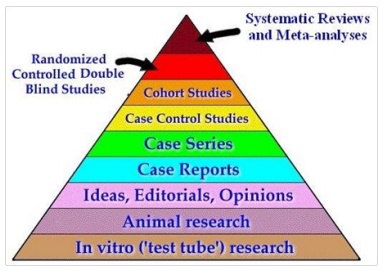
OBJECTIVES AND STUDY DESIGN